Trial Data / Platform Tour
Help Index > Trial Data / Platform Tour
1. Background
Patient-based data from clinical trials cannot generally be made public for data protection reasons. Since there is no concept and few standards for data
sharing, a lot of data remains hidden forever.
2. Solution
Data in the Health Atlas is systematically prepared together with the data generating groups and then uploaded and presented in various formats and
for online exploring. The metadata enriched CDISC ODM format is used consistently, SPSS and Excel formats are generated.
The Health Atlas is bound by data protection laws and patient approvals. Therefore, only basic results of the studies are offered to registered and
authorized users online, which is decided by the study management. Metadata is public.
3. Guided Tour
Task: Retrieve and view data from Trials of the LIFE Head and Neck Cancer Group (LIFE-HNC)

I2b2 queries can be much more complex. A complete introduction to the i2b2 system goes beyond the scope of this tour. Besides the included “Help”-Button on the upper right, there a many public i2b2-manuals available; e.g from Boston University:
Which projects are related to head and neck cancer?
- Type “head and neck cancer” into the search bar or go to Human Diseases of the Health Atlas and select “head and neck cancer” in the tree.
- Select the project “LIFE Head and Neck Cancer Group”
- You are now here: https://health-atlas.de/lha/7Q0CR2P3M0-6.
Which trials are available from the LIFE-HNC?
- Scroll down to “Related items”
- Click on “Stratification of head and neck squamous cell carcinomas (HNSCC)” in the “Studies” section; you are now here: https://health-atlas.de/lha/803RKY0RUT-6
Download studie data and metadata of the presented studie
- navigate through the tree by double clicking the data files
Explore Data
- You need user credentials to explore contents from this project (which are: username: lha, password: lha)
- Click on the i2b2 button of one of the data files
- Your are now redirected to the Health Atlas instance of the i2b2 Query and Analysis Tool from the i2b2/tranSMART Foundation (see https://www.i2b2.org/)
- The login form should be pre-filled, if not, use the credentials mentioned above.
- You have now access to all published trial-data of this group.
- In i2b2, look into upper left “Navigate Terms” click on “Trials”. This opens all available trial data sets.
- The number to the right (e.g. 124) indicates how many patients own this “concept”
- Open “Demography/Sex"; drag the concept “male” to “Group 1” on the right
- Open “HPV status” and “HPV16 DNA RNA status (code)”; drag “DNA+RNA-” to “Group 2” on the right
- Click “Run Query”; check “Age patient breakdown” (scroll a little)
- Click “Ok”; your just performed an sql join statement…
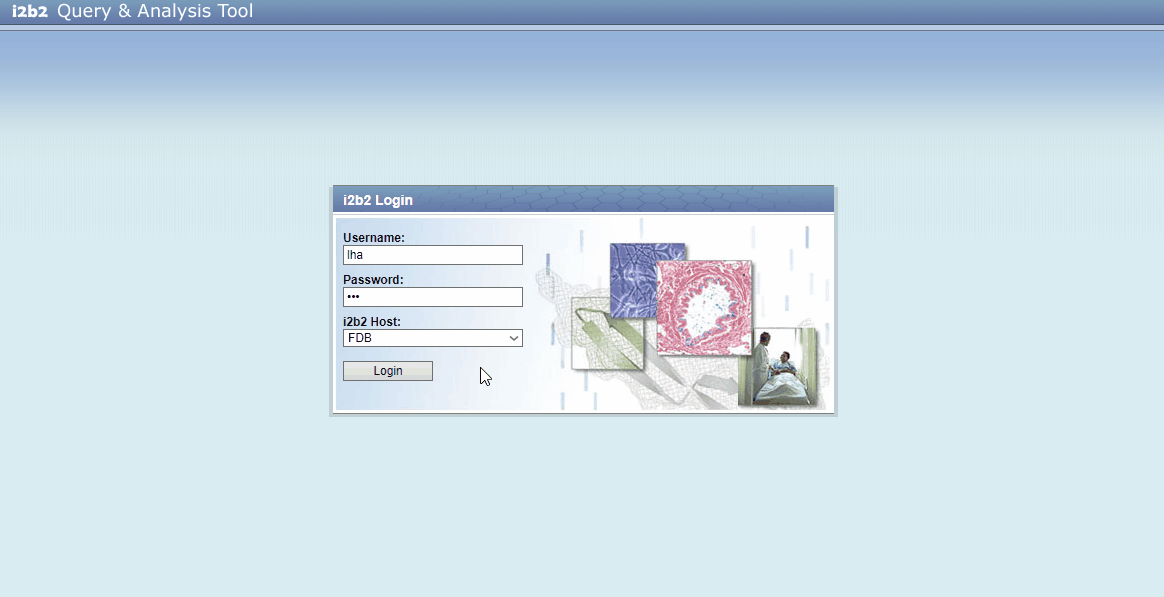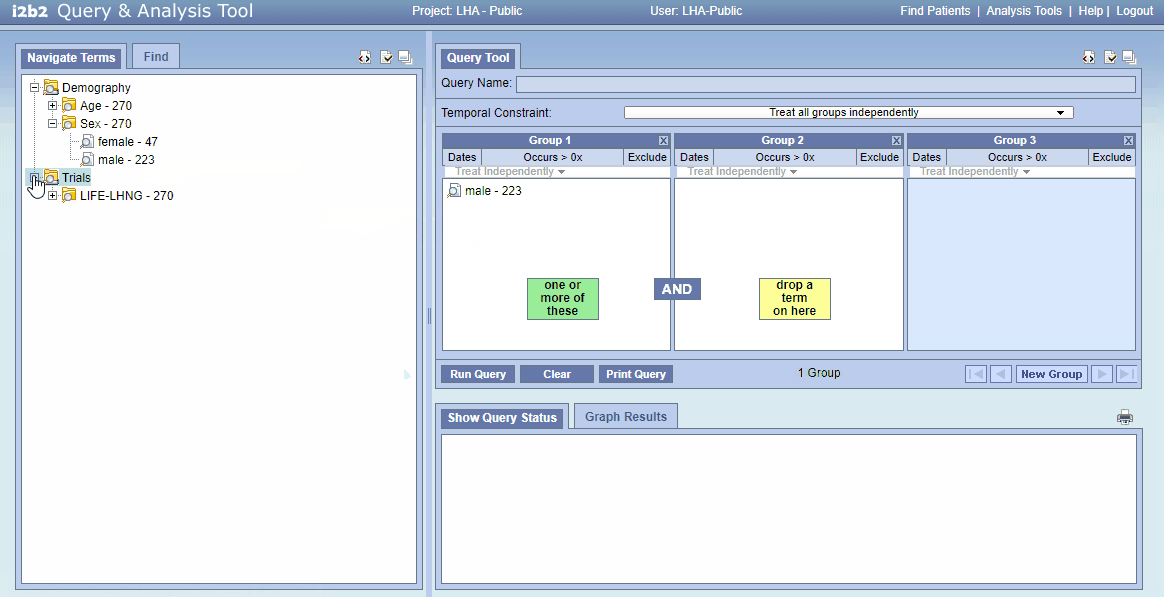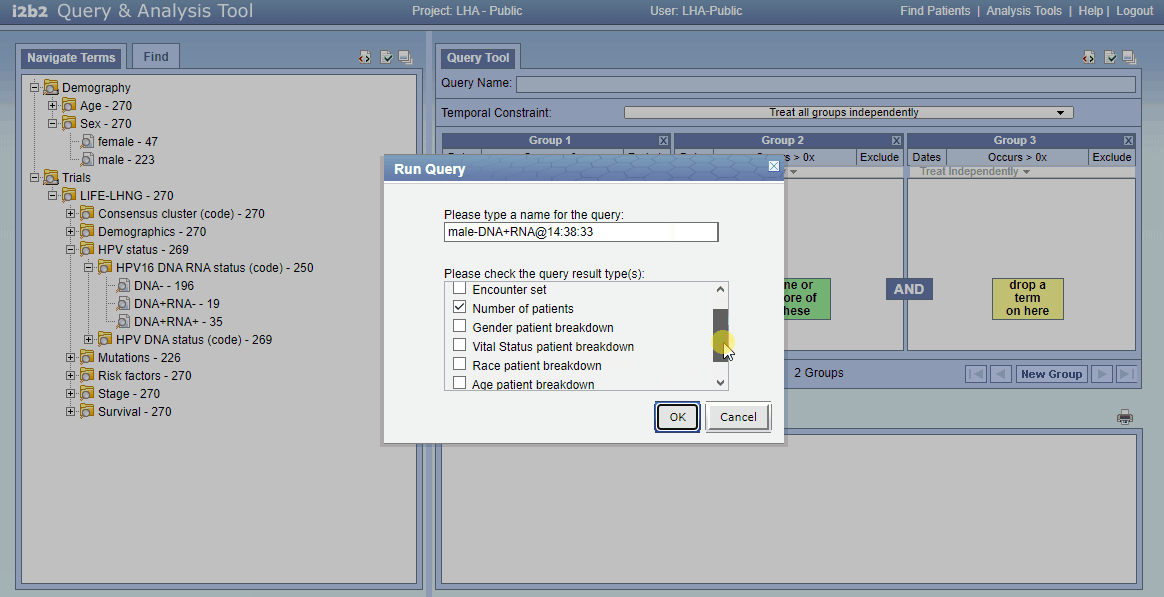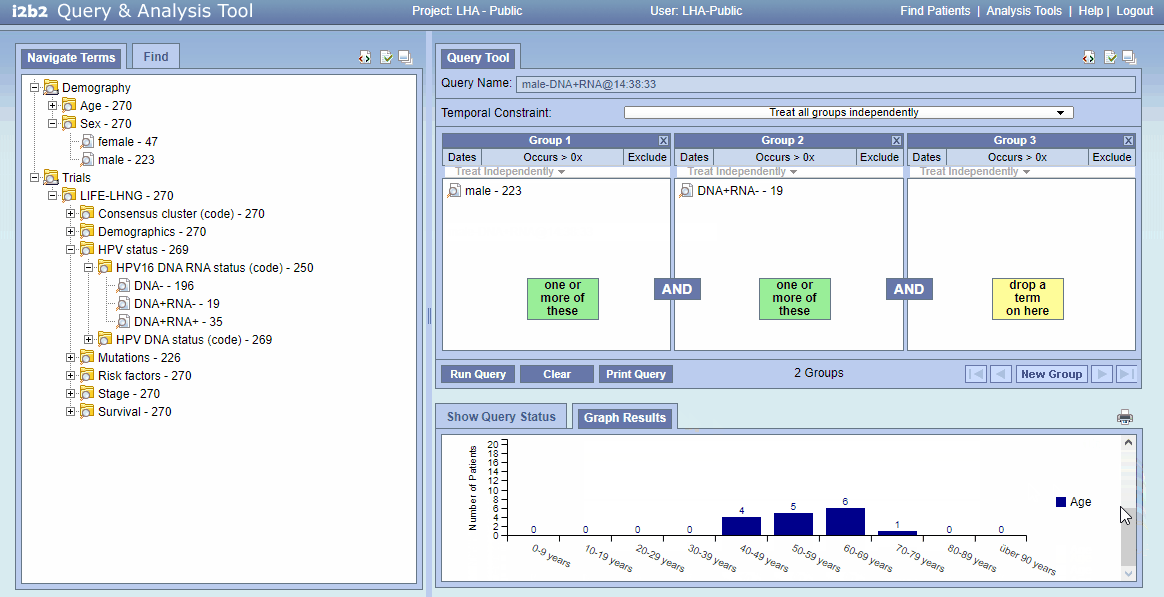
- See result in in “Graph Results”: 16 patients are found. For age distribution scroll “Graph Results” down a little
I2b2 queries can be much more complex. A complete introduction to the i2b2 system goes beyond the scope of this tour. Besides the included “Help”-Button on the upper right, there a many public i2b2-manuals available; e.g from Boston University:
- http://cri.uchicago.edu/wp-content/uploads/2015/02/i2b2_quick_start_v1.pdf a nice pdf manual
- http://sites.bu.edu/bu-i2b2/intro-to-i2b2/ a nice web intro